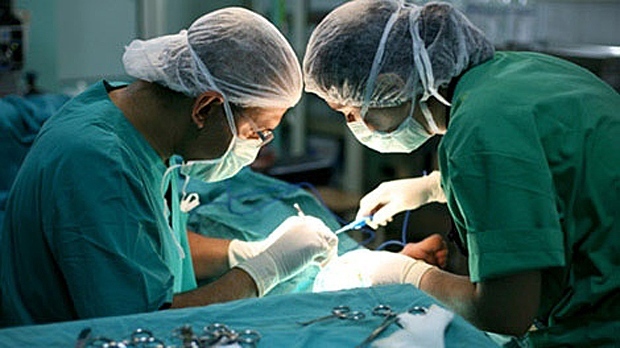
TORONTO -- Despite a growing number of lawsuits linked to the use of vaginal mesh implants, the proportion of women who need reparative surgery for complications from the device is relatively low -- at least for those treated for stress incontinence, an Ontario study suggests.
A study by the Institute for Clinical Evaluative Sciences (ICES) found that about three per cent of women treated with a transvaginal mesh sling for stress incontinence -- the kind of urine leakage typically brought on by laughing, coughing, sneezing or exercise -- have needed surgery to remove or alter the implant.
"Our study shows that, after 10 years of followup, one in 30 Ontario women required mesh revision or removal surgery," said principal researcher Dr. Blayne Welk, a urologist at St. Joseph's Hospital in London, Ont., and an adjunct scientist at ICES.
Vaginal mesh implants are made from a polymer plastic and work by supporting the urethra, the tube through which urine flows, he said. "When there's activity, the urethra can't move and urine can't leak out."
The implants, the most common surgical intervention for female stress incontinence over the past 20 years, are also used to treat pelvic organ prolapse (POP), in which the bladder and uterus have begun to sag.
But in some cases, the slings can cause complications, such as chronic pelvic, genital or groin pain and an inability to properly urinate. Among the most serious adverse effects are infection, tissue erosion and organ perforation.
The 2002-2012 study, published Wednesday in the journal JAMA Surgery, examined health records for almost 60,000 Ontario women who received a vaginal mesh implant for stress incontinence. Researchers found that 1,307 were required to have the device removed or altered in some way as a result of complications.
Welk said the study showed that surgeons who performed a lower number of implants had a 37 per cent increased likelihood of a patient having complications, compared to their higher-volume counterparts.
But he said he was heartened to see the rate of removal/revision surgery was less than three per cent.
"As a surgeon, I find it reassuring that the rate's relatively low, and I think the most important thing that statistic does is help us educate our patients on the risk of this surgery. And it reaffirms the fact that complications are rare, but they're not unheard of. They do happen with this procedure."
Paul Miller, a partner in the Toronto law firm Will Davidson LLP, said he was surprised at the study's one-in-30 finding, given that thousands of women in Canada and the United States have been or are plaintiffs in class-action and individual lawsuits against manufacturers of the mesh slings.
"I'm surprised it was such a low number, actually," Miller said Wednesday. "I was surprised considering the number of clients."
His firm is representing about 225 women, who overall are suing seven different manufacturers of the devices. About 35 cases against the company American Medical Systems were settled out of court in December for an undisclosed amount. That company is also facing a class-action lawsuit in Canada.
Miller said an estimated 5,000 to 7,000 Canadian women who had vaginal mesh implants for stress urinary incontinence or pelvic organ prolapse are plaintiffs in lawsuits due to complications.
In the U.S., 50,000 to 70,000 women fitted with the slings have also brought suit against manufacturers, which include Johnson and Johnson, Ethicon, Boston Scientific and Atrium.
"One of the things I hear from women all the time -- and it's the saddest thing -- is that 'the doctor says it's in my head. It's not the mesh. There's no problem with the mesh,"' said Miller.
"That is a problem that women are encountering."
The US. Food and Drug Administration and Health Canada have both issued warnings about the risk of complications from mesh slings, including painful intercourse and urination, vaginal and urinary tract infections, and injuries to the bladder, bowel and vagina.
In 2011, the FDA strengthened its warning, saying serious complications associated with surgical mesh for transvaginal repair of POP are not rare. The agency also said vaginal mesh procedures do not necessarily improve quality of life for patients over standard surgeries and instead can expose them to greater risk.
The ICES researchers say their findings support the regulatory agencies' statements suggesting that patients should be counselled that serious complications can occur with mesh-based stress urinary incontinence procedures and that surgeons should achieve expertise in their chosen procedure.
"The most important thing for women to realize is they should talk to their surgeon about their experience and their background with the product that they're using and make sure they're happy with their surgeon's ability to manage potential complications, if they should come up," said Welk.