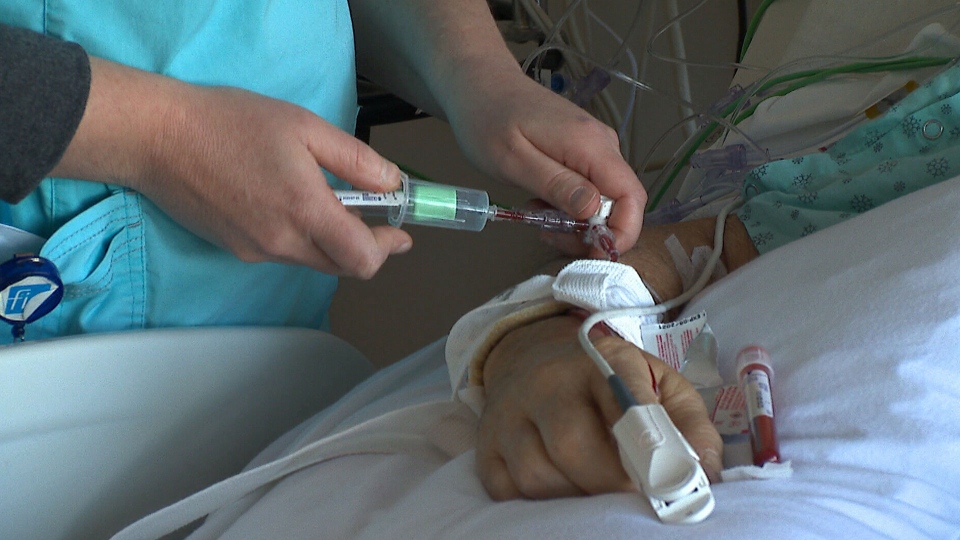
LONDON, ONT. -- Clinical trials involving a human protein and its effects as a potential therapy for COVID-19 patients with sepsis are now underway at Lawson Health Research Institute in London, Ont.
The protein annexin A5 has been found to have anti-coagulation and anti-inflammatory properties and could benefit sepsis patients.
Sepsis is a potentially deadly condition that happens when the body's response to an infection damages major organs and tissues. Many severe COVID-19 patients develop sepsis.
“When the pandemic started, there was no proven treatment for sepsis, including sepsis as a result of COVID-19. Based on clinical trials during the pandemic, we now use steroids and other treatments to try to help, but the results and effects aren’t dramatic and we see patients who have these treatments and still progress and end up in the ICU,” says Dr. Claudio Martin, associate scientist at Lawson and Intensive Care Physician at LHSC.
A research team at Lawson has found that annexin A5 can help prevent inflammation and improve organ function in animals. It's now moved to a clinical trial with critically ill COVID-19 patients, using a manufactured form of the protein.
Enrollment for the trial is underway with the hopes of having 60 people in the initial study. If successful, it will expand beyond COVID-19 patients with sepsis, but to other sepsis patients also.
“If in fact Annexin A5 is shown to be effective in sepsis, then this will be a huge benefit for society because sepsis is the leading cause of death worldwide,” says Dr. Qingping Feng, Lawson scientist.