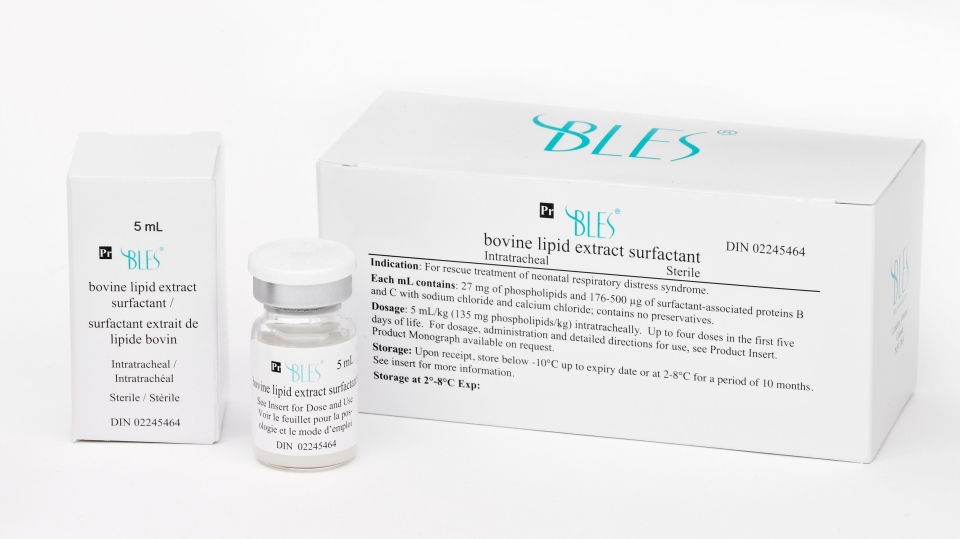
LONDON, ONT. -- It’s called BLES, a therapeutic treatment created in London years ago that was first used to improve lung function in pre-term babies.
“BLES is something that pretty much routinely now pre-term babies born less than 32 weeks or 30 weeks receive prophylacticaly so they can breath on their own without the need of mechanical ventilation,” says Dr. Jim Lewis at the Lawson Health Research Institute.
BLES uses what’s called surfactant, which is made out of lipids and proteins in the body, and helps with proper lung function.
Not only is it successfully used in pre-term babies, researchers have found benefits for patients with acute respiratory distress symptom (ARDS).
The research team has now turned to patients who are experiencing severe respiratory issues due to COVID-19, which reduces the amount of surfactant in the lungs making it harder to breathe.
“Those who do require ventilator support, we know that the ventilation itself can damage surfactant, so it’s a double-edged sword where the ventilator is needed to survive, but it can also propagate the injury on top of infection of the virus,” Lewis says.
And that can then lead to further complications.
“This process continues and damages more and more areas and the patient gets worse and worse,” says Dr. Ruud Veldhuizen, a Lawson scientiest. “So surfactant, by opening up the areas that are not functioning, may actually benefitting those patients.”
That’s why the research team is conducting a study using surfactant on COVID-19 patients requiring ventilation to see if this drug will improve and reduce the amount of ventilation needed.
“We feel it will it will not only prevent the damaging effects of the ventilator itself, but also because surfactant as a lipoprotein complex has anti-inflammatory properties, there’s an added benefit in giving surfactant early on in this disease so we can decrease this big inflammatory response that these patients exhibit,” says Lewis.
If proven successful the next steps would be to launch a larger study with the hopes this treatment could become a life-saving option for severe COVID-19 patients.